COVID-19 Variant ID | For Health Departments & Hospitals
MicroGenDX whole genome sequencing identifies known SARS-CoV-2 variants
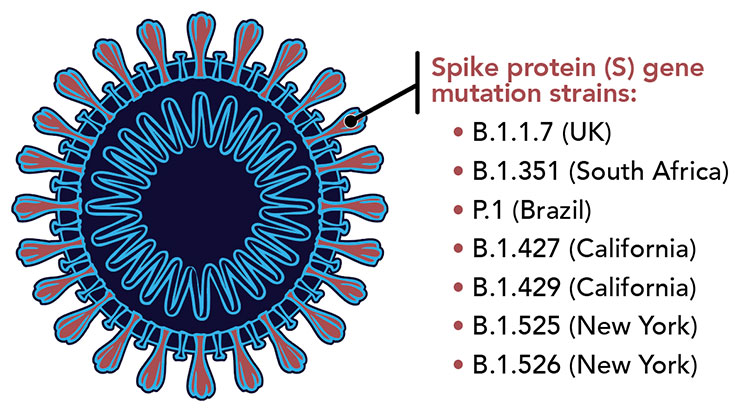
MicroGenDX, the industry leader in providing Next Generation DNA Sequencing (NGS) for clinical diagnostics, now offers whole genome sequencing for identifying known variants of SARS-CoV-2 described by the CDC as epidemiologically significant. Currently, identified variants do not require different therapeutic or infectious prevention approaches. But with the emergence of new variants, whole genome testing and research of SARS-CoV-2 may assist with discovering and describing new management approaches. Researchers have already concluded that variants carrying the E484K mutation may escape immune response in some vaccinated individuals.
Impact on health departments
New variants continue to emerge rapidly around the globe, causing sequencing efforts at health departments and universities to surveil a small portion of the population, and in some cases limit their sequencing efforts to breakthrough cases. MicroGenDX is proud to offer the first commercially available, quick turnaround, economical, and accurate option for SARS-CoV-2 whole genome sequencing—to combat the spread of emerging variants and gain a better understanding of the epidemiology of SARS-CoV-2.
Contact customer service or call 855-208-0019 to get started or for more information.
COVID-19 Variant ID FAQ
COVID-19 Variant ID FAQ
Please note: This assay is intended to be used as an epidemiological tool and is not intended for diagnosis or for guiding therapeutic management.
References
- Xie X, Liu Y, Liu J, et al. Neutralization of SARS-CoV-2 spike 69/70 deletion, E484K and N501Y variants by BNT162b2 vaccine-elicited sera. Nature Medicine. 2021 Feb 8. doi: 10.1038/s41591-021-01270-4
